生化分析试剂 Biochemical Assay Reagents
Imidazole;(Synonyms: 咪唑; 1,3-Diaza-2,4-cyclopentadiene; Glyoxaline) 纯度: 99.86%
Imidazole是一种平面五元环, 是一种高极性化合物,已被广泛用作腐蚀抑制剂。
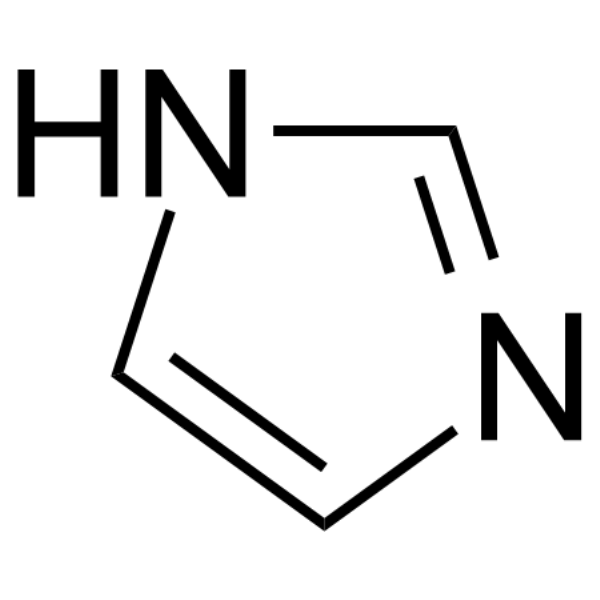
Imidazole Chemical Structure
CAS No. : 288-32-4
| 规格 | 价格 | 是否有货 | 数量 |
|---|---|---|---|
| Free Sample (0.1-0.5 mg) | ; | Apply now | ; |
| 10;mM;*;1 mL in Water | ¥500 | In-stock | |
| 500 mg | ¥400 | In-stock | |
| 5 g | ¥500 | In-stock | |
| 10 g | ; | 询价 | ; |
| 50 g | ; | 询价 | ; |
* Please select Quantity before adding items.
Imidazole 相关产品
bull;相关化合物库:
- Natural Product Library Plus
- Bioactive Compound Library Plus
- Metabolism/Protease Compound Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Fragment Library
- FDA Approved amp; Pharmacopeial Drug Library
- Anti-Blood Cancer Compound Library
- Microbial Metabolite Library
- Food-Sourced Compound Library
| 生物活性 |
Imidazole is a planar 5-membered ring. Imidazole is a highly polar compound. Imidazole has been used extensively as a corrosion inhibitor. Imidazole is incorporated into many important biological molecules. The most pervasive is the amino acid histidine, which has an imidazole side-chain. Imidazole is useful as a buffer in the pH range of 6.2-7.8 One of the applications of imidazole is in the purification of His-tagged proteins in immobilised metal affinity chromatography(IMAC). Imidazole is used to elute tagged proteins bound to Ni ions attached to the surface of beads in the chromatography column. An excess of imidazole is passed through the column, which displaces the His-tag from nickel co-ordination, freeing the His-tagged proteins. Imidazole has become an important part of many pharmaceuticals. Synthetic imidazoles are present in many fungicides and antifungal, antiprotozoal, and antihypertensive medications. Imidazole is part of the theophylline molecule, found in tea leaves and coffee beans, which stimulates the central nervous system. It is present in the anticancer medication mercaptopurine, which combats leukemia by interfering with DNA activities. |
||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 分子量 |
68.08 |
||||||||||||||||
| Formula |
C3H4N2 |
||||||||||||||||
| CAS 号 |
288-32-4 |
||||||||||||||||
| 中文名称 |
咪唑;异吡唑;间二氮茂 |
||||||||||||||||
| 运输条件 |
Room temperature in continental US; may vary elsewhere. |
||||||||||||||||
| 储存方式 |
|
||||||||||||||||
| 溶解性数据 |
In Vitro:;
H2O : ≥ 100 mg/mL (1468.86 mM) * “≥” means soluble, but saturation unknown. 配制储备液
*
请根据产品在不同溶剂中的溶解度选择合适的溶剂配制储备液;一旦配成溶液,请分装保存,避免反复冻融造成的产品失效。 |
